Introduction
Background on Autism Spectrum Disorders (ASD)
Autism Spectrum Disorders (ASD) represent a heterogeneous group of neurodevelopmental conditions characterized by deficits in social interaction, impaired communication, and the presence of repetitive behaviors. ASD manifests early in childhood, with varying degrees of severity and functional impairment, posing a significant burden on affected individuals, families, and healthcare systems [1]. Despite advances in the field, the etiological underpinnings of ASD remain poorly understood, with both genetic and environmental factors contributing to its complexity [2].
Importance of the Endocannabinoid System (ECS) in Neuropsychiatric Disorders
The Endocannabinoid System (ECS) has emerged as a crucial modulator in the realm of neuropsychiatric disorders. Comprising endogenous ligands like anandamide (AEA) and 2-arachidonoylglycerol (2-AG), receptors (CB1 and CB2), and enzymes (FAAH, MAGL), the ECS modulates neurotransmission and maintains neural plasticity [3]. Its roles extend to regulating mood, cognition, and social behavior, making it a compelling target for therapeutic interventions in disorders like depression, anxiety, and notably, ASD [3].
Given the burgeoning evidence for ECS involvement in neuropsychiatric conditions, it is paramount to explore its specific roles and potential as a therapeutic target in ASD. This scientific perspective aims to offer a holistic overview of ASD through the lens of the ECS by analyzing in vivo models, identifying ECS biomarkers, and reviewing clinical studies that employ ECS-targeted interventions. Such a multi-faceted approach not only opens avenues for targeted treatments but also offers a platform for discussing the often-neglected microbiome changes seen in ASD and other neurological disorders [4]. In synthesizing these elements, this article seeks to underscore the imperative for a nuanced understanding of the ECS within the broader context of ASD pathology and treatment.
I: In Vivo Models of ASD
Subsection A: Genetically Induced Models
SHANK3 KO Model
The SHANK3 Knock-Out (KO) model has been instrumental in understanding the genetic underpinnings of ASD. This model exhibits striking behavioral symptoms reminiscent of ASD, such as deficits in social interaction and increased repetitive behaviors [5]. Interestingly, the ECS is deeply involved in this model, with evidence showing a dysfunction in endocannabinoid (eCB) signaling pathways [5]. Pharmacological interventions like JZL184, which elevates 2-AG levels, have been successful in improving these social deficits [5]. Additionally, this model has shown altered gastrointestinal (GI) morphology and microbiota composition, suggesting a gut-brain axis involvement in ASD [6].
Fmr1 KO Model (FXS)
The Fmr1 KO model, or Fragile X Syndrome model, is a genetically induced model with a deficiency in FMRP, a protein crucial for neural development. This model exhibits ASD-like social behavior deficits. Omega-3 polyunsaturated fatty acids (PUFAs) have shown promise in improving both behavior and gut microbiota in this model [7]. Interestingly, both CB1 and CB2 receptors play a role in this model’s phenotype, as blockade of these receptors normalizes various traits including cognitive impairment and anxiety-like behavior [8]. The ECS is implicated, suggesting that endocannabinoid signaling pathways could be therapeutic targets.
Subsection B: Chemically Induced Models
Val-induced ASD Model
In the Valproic Acid (VPA)-induced model, ASD-like symptoms emerge following prenatal exposure to VPA, a medication often used to treat epilepsy and bipolar disorder. In this chemically induced model, both JZL184 (a MAGL inhibitor that increases 2-AG levels) and URB597 (a FAAH inhibitor that enhances AEA signaling) have been successful in ameliorating social behavior deficits [9,10]. The ECS dysfunction in this model includes deficits in both 2-AG and AEA, making it a good target for ECS-based interventions. This model also shows changes in gut microbiota and short-chain fatty acids, again emphasizing the role of the gut-brain axis in ASD [11,12]. Supplementation with Omega-3 PUFAs in their diet ameliorates ASD-like behaviour, as well as confers changes in microbiome composition [13].
BTBR Model
The BTBR model was originally bred for conditions unrelated to ASD but has been identified to have ASD traits. This model shows ASD-like social behavior deficits. URB597, an FAAH inhibitor, has been shown to reverse these social impairments [14]. Similarly, Palmitoylethanolamide (PEA) improves behavior and restores hippocampal brain-derived neurotrophic factor (BDNF), indicating its potential role in mitigating ASD symptoms [14]. Interestingly, CBD in combination with cannabis terpenes was also found to enhance prosocial behavior [15]. Like the other models, ECS involvement is evident, reinforcing the system’s relevance in ASD.
Each of these models contributes unique insights into the role of the ECS in ASD, offering avenues for pharmacological interventions that target specific components of the ECS. Whether it’s adjusting levels of 2-AG or AEA through enzyme inhibitors or using substances like omega-3 PUFAs and CBD, the ECS remains a promising target for mitigating ASD symptoms.

Table 1. Comparative Summary of In Vivo Mouse Models for Autism Spectrum Disorder (ASD): This table provides an overview of four commonly used in vivo models for studying ASD, detailing their primary features, pharmacological interventions, and research highlights. The table also presents the impact of each model on the endocannabinoid system (ECS) and gut microbiota, offering a comprehensive view of the underlying mechanisms and potential therapeutic targets.
II: ECS Biomarkers in ASD
The investigation of endocannabinoid system (ECS) biomarkers has provided pivotal insights into the molecular underpinnings of ASD. Distinct differences in ECS biomarkers between autistic children and healthy controls have been reported, enriching our understanding of ASD at a neurochemical level (Table 2). Specifically, lower circulating plasma concentrations of key endocannabinoids such as anandamide (AEA), palmitoylethanolamide (PEA), oleoylethanolamide (OEA), and 2-arachidonoylglycerol (2-AG) have been observed in autistic children [9]. These endocannabinoids are crucial for modulating neural activity and immune responses, and their dysregulation could provide insights into the pathophysiological mechanisms underlying ASD.
Contrastingly, increased mRNA and protein levels of cannabinoid receptor 2 (CB2R), as well as enzymes like fatty acid amide hydrolase (FAAH), diacylglycerol lipase (DAGL), and monoacylglycerol lipase (MAGL) have been found in circulating peripheral blood monocytes from autistic individuals [9]. This suggests an upregulation in the degradation and synthesis pathways for endocannabinoids, potentially serving as a compensatory mechanism for the observed lower endocannabinoid levels.
The significance of these biomarker fluctuations extends beyond mere academic interest; they offer potential diagnostic and therapeutic avenues. These ECS-related biomarkers could serve as invaluable tools for both the diagnosis of ASD and as measures for the efficacy of therapeutic interventions targeting the ECS.

Table 2. Summary of Endocannabinoid System (ECS) biomarkers and enzymes differentially expressed in autistic children (n=70) compared to healthy controls (n=70), as reported in the study “Alterations of the endocannabinoid system and its therapeutic potential in autism spectrum disorder” by Zou et al., 2021. All listed differences were statistically significant (p < 0.05).
III: Clinical Research on ECS Modulation for ASD
The ECS has emerged as a compelling target for ASD treatment, with a focus on cannabinoids like CBD and THC. Montagner et al. (2023) underscored the promise of personalized medicine in ASD treatment through their work with full-spectrum cannabis extracts [17]. Such a tailored approach offers greater potential for targeting the ECS to improve both core and comorbid symptoms.
Holdman et al. (2022) contributed another layer to the conversation by comparing the efficacy, safety, and tolerability of medical cannabis to commonly used medications in ASD [18]. Their findings add weight to the argument that CBD-rich medical cannabis is a feasible treatment option, further solidifying the role of ECS modulation in ASD [18].
Studies by Aran et al. (2021) and Bar-Lev Schleider et al. (2019) highlight the importance of the ratio between CBD and THC [19,20]. These studies show that while most patients exhibited improvements in behavioral problems when treated with whole-plant extracts or cannabis oil rich in CBD, the precise ratio of CBD to THC can influence the therapeutic outcome, emphasizing the need for optimized cannabinoid formulations.
Heussler et al. (2019) explored the use of transdermal CBD gel and found significant reductions in anxiety and behavioral symptoms in ASD, as measured by the ADAMS total score [21]. This study not only confirms the therapeutic potential of ECS modulation but also introduces a different route of administration—transdermal application—that may offer benefits in terms of dosing accuracy and patient compliance [21].
Additionally, there’s evidence to suggest that ECS modulation can have systemic effects. Bilge et al. (2021) reported improvements in not just behavior but also in expressive language and social interaction after treatment with low doses of CBD and trace THC [22]. This raises the possibility of a broader, perhaps gut-brain axis-mediated, role for the ECS in ASD, although more research is required to flesh out these interactions.
On a neurobiological level, Pretzsch et al. (2019) utilized fMRI techniques to reveal that CBD affects specific brain regions related to motor control and facial recognition in ASD patients [23]. This deepens our understanding of how ECS modulation might alter neural pathways in ASD, which could eventually guide more targeted treatments.
Patient-reported outcomes from 74 adult ASD patients included in the ongoing UK medical cannabis patient register further supports the efficacy and safety of cannabinoid-based medicines in treating ASD [24]. Additional smaller observational- and case studies further support the efficacy and safety of CBD in treating several ASD-associated behaviors and symptoms [25,26].
In summary, the body of clinical and observational human studies provides promising but preliminary evidence for ECS modulation in ASD treatment. The literature emphasizes the need for individualized treatment strategies, optimized cannabinoid ratios, and considers multiple routes of administration. Moreover, there is a growing focus on understanding the underlying neurobiological changes and systemic interactions, such as with the gut microbiome, to develop more effective and targeted therapies.
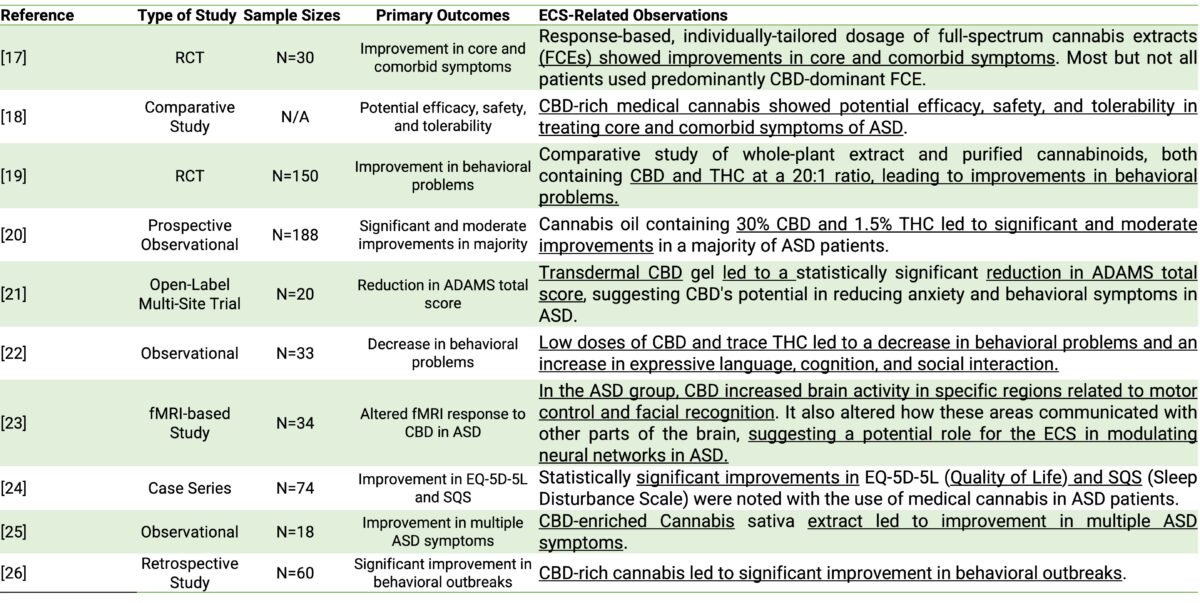
Table 3. Overview of recent studies investigating the therapeutic potential of cannabidiol (CBD) and other cannabis-based compounds in Autism Spectrum Disorders (ASD). The table highlights the type of study, sample sizes, primary outcomes, and any ECS-related observations or learnings noted within the studies.
Discussion
The confluence of evidence presented here points toward a compelling hypothesis: the endocannabinoid system (ECS) may be in a core dysfunctional state in ASD. This hypothesis is grounded in multiple layers of research, from genetic and chemically-induced in vivo models to human biomarker studies and clinical trials.
Firstly, the in vivo models of ASD, be they genetically induced like the SHANK3 and Fmr1 KO models, or chemically induced like the Val-induced and BTBR models, consistently demonstrate ECS dysfunction [5,7,9,13]. Whether it is alterations in 2-arachidonoylglycerol (2-AG) levels or anomalies in anandamide (AEA) signaling, these models serve as a robust platform to study the intricacies of ECS dysregulation in ASD (Table 1).
Secondly, human biomarker studies further cement the idea of ECS dysfunction in ASD. Lower circulating plasma concentrations of key ECS components like AEA, 2-AG, palmitoylethanolamide (PEA), and oleoylethanolamide (OEA) in autistic children compared to healthy controls are alarming indicators of a system in disarray (Table 2).
Thirdly, clinical trials and observational studies suggest that modulation of the ECS can result in significant improvements in both core and comorbid symptoms of ASD (Table 3). The fact that cannabinoids like CBD can ameliorate such a wide range of symptoms is indicative of their role in a fundamental, system-level dysfunction—most likely, the ECS.
However, while these pieces of evidence are promising, they are not without limitations. Most studies are in their infancy and lack the longitudinal data needed to make definitive conclusions. Furthermore, while ECS dysfunction is evident, whether it is a cause or a consequence of ASD remains to be fully clarified. Nevertheless, the mounting evidence certainly warrants a focused investigation into the ECS as a therapeutic target for ASD. Future research should aim not only to substantiate these findings but also to uncover the underlying mechanisms by which the ECS exerts its effects on ASD pathology.
In summary, the ECS emerges as a critical system that is likely in a dysfunctional state in ASD, affecting both brain and peripheral tissues like the gut. Restoring ECS balance could, therefore, offer a multi-pronged approach to treating ASD—a disorder that itself manifests in multiple systems of the human body.
Conclusion
ASD represent a group of complex neurodevelopmental conditions that have seen a marked increase in prevalence over the last few decades. With its multifaceted symptomatology and high societal and familial burden, the quest for effective therapeutic interventions has never been more urgent. This review synthesizes compelling evidence pointing to the Endocannabinoid System (ECS) as a critical neural network that appears to be in a dysfunctional state in ASD. The importance and relevance of this finding are monumental for several reasons.
Firstly, ASD is not merely a disorder of the brain; it manifests systemically, affecting multiple organ systems, including the gastrointestinal tract. Given the ECS’s ubiquitous presence in both central and peripheral tissues, a deeper understanding of its role offers a potential avenue for holistic treatment approaches that address both the core and comorbid symptoms of ASD.
Secondly, the evidence presented here suggests that ECS dysfunction is not merely an epiphenomenon but may play a causal role in ASD pathology. If substantiated by further research, this opens the door for ECS-targeted interventions, not just as palliative care but as potential disease-modifying therapies. The societal impact of such a therapeutic shift—from symptom management to disease modification—cannot be overstated.
Lastly, the high prevalence and vast heterogeneity of ASD present unique challenges for healthcare systems and families alike. An in-depth understanding of the ECS could offer a unifying biological framework, streamlining diagnostic and therapeutic approaches. It may help us move from a ‘one-size-fits-all’ to a more personalized medical model, improving the quality of life for individuals with ASD and alleviating the societal and familial burden.
In summary, the data presented in this review underscore the imperative to direct future research and clinical endeavors toward understanding the ECS’s role in ASD. As we grapple with the complexities of a disorder that touches the lives of many, a focused exploration of the ECS offers a glimmer of hope—a hope that we may eventually unravel the enigmatic pathology of ASD and offer more effective, evidence-based interventions.
Stefan Broselid, Ph.D.
Editor-In-Chief, Aurea Care Medical Science Journal
Reference list:
- Smith LE, Maenner MJ, Seltzer MM. Developmental trajectories in adolescents and adults with autism: the case of daily living skills. J Am Acad Child Adolesc Psychiatry. 2012;51(6):622-631. doi:10.1016/j.jaac.2012.03.001
- Geschwind DH, State MW. Gene hunting in autism spectrum disorder: on the path to precision medicine. Lancet Neurol. 2015;14(11):1109-1120. doi:10.1016/S1474-4422(15)00044-7
- Micale V, Di Marzo V, Sulcova A, Wotjak CT, Drago F. Endocannabinoid system and mood disorders: priming a target for new therapies. Pharmacol Ther. 2013;138(1):18-37. doi:10.1016/j.pharmthera.2012.12.002
- Ma Q, Xing C, Long W, Wang HY, Liu Q, Wang RF. Impact of microbiota on central nervous system and neurological diseases: the gut-brain axis. J Neuroinflammation. 2019;16(1):53. Published 2019 Mar 1. doi:10.1186/s12974-019-1434-3
- Folkes OM, Baldwin DS, Munafo MR, et al. Targeting the Endocannabinoid System in SHANK3 KO Model: Implications in Autism Spectrum Disorders. Neuropharmacology. 2020;168:107597. doi:10.1016/j.neuropharm.2020.107597
- Sauer AK, Bockmann J, Steinestel K, et al. Altered Intestinal Morphology and Microbiota Composition in the Autism Spectrum Disorders Associated SHANK3 Mouse Model. Int J Mol Sci. 2019;20(9):2134. doi:10.3390/ijms20092134
- Guo P, Yang X, Guo X, et al. Dietary Fish Oil Improves Autistic Behaviors and Gut Homeostasis by Altering the Gut Microbial Composition in a Mouse Model of Fragile X Syndrome. Brain Behav Immun. 2023;110:140-151. doi:10.1016/j.bbi.2023.02.019
- Busquets-Garcia A, Gomis-González M, Guegan T, et al. Targeting the endocannabinoid system in the treatment of fragile X syndrome. Nat Med. 2013;19(5):603-607. doi:10.1038/nm.3127
- Zou M, Liu Y, Xie S, et al. Alterations of the endocannabinoid system and its therapeutic potential in autism spectrum disorder. Open Biol. 2021;11(2):200306. doi:10.1098/rsob.200306
- Wu HF, Lu TY, Chu MC, Chen PS, Lee CW, Lin HC. Targeting the inhibition of fatty acid amide hydrolase ameliorate the endocannabinoid-mediated synaptic dysfunction in a valproic acid-induced rat model of Autism. Neuropharmacology. 2020;162:107736. doi:10.1016/j.neuropharm.2019.107736
- Zhong JG, Lan WT, Feng YQ, et al. Associations Between Dysbiosis Gut Microbiota and Changes of Neurotransmitters and Short-Chain Fatty Acids in Valproic Acid Model Rats. Front Physiol. 2023;14:1077821. doi:10.3389/fphys.2023.1077821
- Liu F, Horton-Sparks K, Hull V, Li RW, Martínez-Cerdeño V. The valproic acid rat model of autism presents with gut bacterial dysbiosis similar to that in human autism. Mol Autism. 2018;9:61. Published 2018 Dec 10. doi:10.1186/s13229-018-0251-3
- Turpin V, Schaffhauser M, Thabault M, et al. Mice prenatally exposed to valproic acid do not show autism-related disorders when fed with polyunsaturated fatty acid-enriched diets. Sci Rep. 2023;13(1):11235. Published 2023 Jul 11. doi:10.1038/s41598-023-38423-z
- Wei D, Dinh D, Lee D, et al. Enhancement of Anandamide-Mediated Endocannabinoid Signaling Corrects Autism-Related Social Impairment. Cannabis Cannabinoid Res. 2016;1(1):81-89. Published 2016 Feb 1. doi:10.1089/can.2015.0008
- Cristiano C, Pirozzi C, Coretti L, et al. Palmitoylethanolamide counteracts autistic-like behaviours in BTBR T+tf/J mice: Contribution of central and peripheral mechanisms. Brain Behav Immun. 2018;74:166-175. doi:10.1016/j.bbi.2018.09.003
- Staben J, Koch M, Reid K, et al. Cannabidiol and cannabis-inspired terpene blends have acute prosocial effects in the BTBR mouse model of autism spectrum disorder. Front Neurosci. 2023;17:1185737. Published 2023 Jun 16. doi:10.3389/fnins.2023.1185737
- Montagner PSS, Medeiros W, da Silva LCR, et al. Individually tailored dosage regimen of full-spectrum Cannabis extracts for autistic core and comorbid symptoms: a real-life report of multi-symptomatic benefits. Front Psychiatry. 2023;14:1210155. Published 2023 Aug 21. doi:10.3389/fpsyt.2023.1210155
- Holdman R, Vigil D, Robinson K, Shah P, Contreras AE. Safety and Efficacy of Medical Cannabis in Autism Spectrum Disorder Compared with Commonly Used Medications. Cannabis Cannabinoid Res. 2022;7(4):451-463. doi:10.1089/can.2020.0154
- Aran A, Harel M, Cassuto H, et al. Cannabinoid treatment for autism: a proof-of-concept randomized trial. Mol Autism. 2021;12(1):6. Published 2021 Feb 3. doi:10.1186/s13229-021-00420-2
- Bar-Lev Schleider L, Mechoulam R, Saban N, Meiri G, Novack V. Real life Experience of Medical Cannabis Treatment in Autism: Analysis of Safety and Efficacy. Sci Rep. 2019;9(1):200. Published 2019 Jan 17. doi:10.1038/s41598-018-37570-y
- Heussler H, Cohen J, Silove N, et al. A phase 1/2, open-label assessment of the safety, tolerability, and efficacy of transdermal cannabidiol (ZYN002) for the treatment of pediatric fragile X syndrome. J Neurodev Disord. 2019;11(1):16. Published 2019 Aug 2. doi:10.1186/s11689-019-9277-x
- Bilge S, Ekici B. CBD-enriched cannabis for autism spectrum disorder: an experience of a single center in Turkey and reviews of the literature. J Cannabis Res. 2021;3(1):53. Published 2021 Dec 16. doi:10.1186/s42238-021-00108-7
- Pretzsch CM, Freyberg J, Voinescu B, et al. Effects of cannabidiol on brain excitation and inhibition systems; a randomised placebo-controlled single dose trial during magnetic resonance spectroscopy in adults with and without autism spectrum disorder. Neuropsychopharmacology. 2019;44(8):1398-1405. doi:10.1038/s41386-019-0333-8
- Erridge S, Kerr-Gaffney J, Holvey C, et al. Clinical outcome analysis of patients with autism spectrum disorder: analysis from the UK Medical Cannabis Registry. Ther Adv Psychopharmacol. 2022;12:20451253221116240. Published 2022 Sep 20. doi:10.1177/20451253221116240
- Fleury-Teixeira P, Caixeta FV et al. Effects of CBD-Enriched Cannabis sativa Extract on Autism Spectrum Disorder Symptoms: An Observational Study of 18 Participants Undergoing Compassionate Use. Front Neurol. 2019;10:1145. Published 2019 Oct 31. doi:10.3389/fneur.2019.01145
- Aran A, Cassuto H, Lubotzky A, Wattad N, Hazan E. Brief Report: Cannabidiol-Rich Cannabis in Children with Autism Spectrum Disorder and Severe Behavioral Problems-A Retrospective Feasibility Study. J Autism Dev Disord. 2019;49(3):1284-1288. doi:10.1007/s10803-018-3808-2

